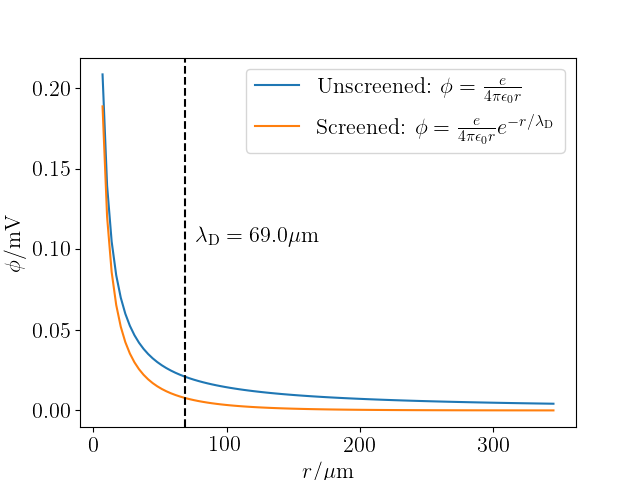
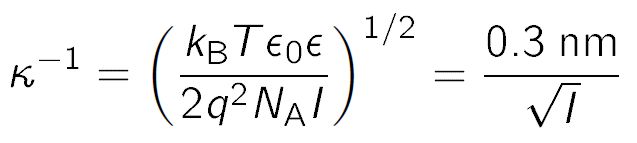Relation between the Debye length, ECV technique coordinate resolution,... | Download Scientific Diagram

nanoHUB.org - Resources: E304 L9.1.4: Nanofluidics - Surface Effects at the Nanoscale: Watch Presentation

SOLVED: Equation to calculate the Debye lengths (λ) in mol/m^3 and 100 mV/m. Use the following aqueous solutions of NaCl at 298 K and discuss how the NaCl concentration influences the Debye

SOLVED: HCl is a covalent molecule. The covalent bond in HCl has 24 % ionic character. The experimental dipole moment of this molecule is found to be 1.50 Debye. Calculate the bond
![SOLVED: Equation List 1. Debye length - 1.304 nm for 1:1 electrolyte [NaCl] - 1.176 nm for 1.2 or 2:1 electrolyte [CaCl2] - 1.152 nm for 2:2 electrolyte [MgSO4] SOLVED: Equation List 1. Debye length - 1.304 nm for 1:1 electrolyte [NaCl] - 1.176 nm for 1.2 or 2:1 electrolyte [CaCl2] - 1.152 nm for 2:2 electrolyte [MgSO4]](https://cdn.numerade.com/ask_images/6fe865672fb949d8969b3329b257473c.jpg)
SOLVED: Equation List 1. Debye length - 1.304 nm for 1:1 electrolyte [NaCl] - 1.176 nm for 1.2 or 2:1 electrolyte [CaCl2] - 1.152 nm for 2:2 electrolyte [MgSO4]

SOLVED: Compute the values of plasma pressure, Debye length, and the number of particles inside the Debye sphere for the following plasmas (n - cm-3, kT - eV). (90 marks) (a) Glow